Dissolved Oxygen: Why It Matters in Aquaculture and Aquariums.
Dissolved oxygen is one of the most critical water quality parameters in aquaculture. Maintaining the correct D.O. levels helps prevent fish stress, encourages healthy growth, and is one of the most important factors in preventing stock losses.
Therefore, understanding how dissolved oxygen behaves — and measuring it correctly — is essential to maintaining healthy aquatic systems.
According to the Global Seafood Alliance, dissolved oxygen is a major concern in aquaculture, as low oxygen levels can quickly stress or kill aquatic species.
Dissolved oxygen refers to the amount of oxygen gas (O₂) that is dissolved in water.
Unlike air, water can only hold a limited amount of oxygen, and that capacity changes constantly depending on environmental conditions. In managed systems such as tanks, ponds, raceways, and aquariums, dissolved oxygen must be actively maintained.
DO is typically measured in milligrams per litre (mg/L).
Safe Dissolved Oxygen Levels
In most aquaculture and aquarium applications:
- 5–14 mg/L is generally considered healthy depending upon the species of fish or aquatic creatures.
- Below ~5 mg/L, many fish species experience stress
- Below ~3 mg/L, serious risk of mortality occurs
Some species require higher minimum levels, for example trout.
Warm-water species may tolerate lower levels — but only briefly
The key point is not a single number, but consistency and trend.
Why Dissolved Oxygen Levels Fluctuate.
Dissolved oxygen levels in water are not constant. They change continuously depending on environmental conditions and system design. The most important factors affecting oxygen levels are temperature, atmospheric pressure, salinity, and water movement.
Temperature
Temperature has one of the strongest effects on dissolved oxygen.
As water temperature increases, its ability to hold oxygen decreases. At the same time, fish and microorganisms become more active and consume oxygen faster.
This combination makes warm weather one of the highest-risk periods for oxygen depletion in:
- Aquaculture ponds
- Outdoor tanks
- Recirculating systems with high stocking density
For example, cool water can hold significantly more oxygen than warm water.
| Water Temperature | Maximum Oxygen Solubility* |
|---|---|
| 5°C | ~12.8 mg/L |
| 10°C | ~11.3 mg/L |
| 20°C | ~9.1 mg/L |
| 30°C | ~7.6 mg/L |
*Approximate values for freshwater at sea level.
This is why oxygen shortages often occur during hot summer conditions.
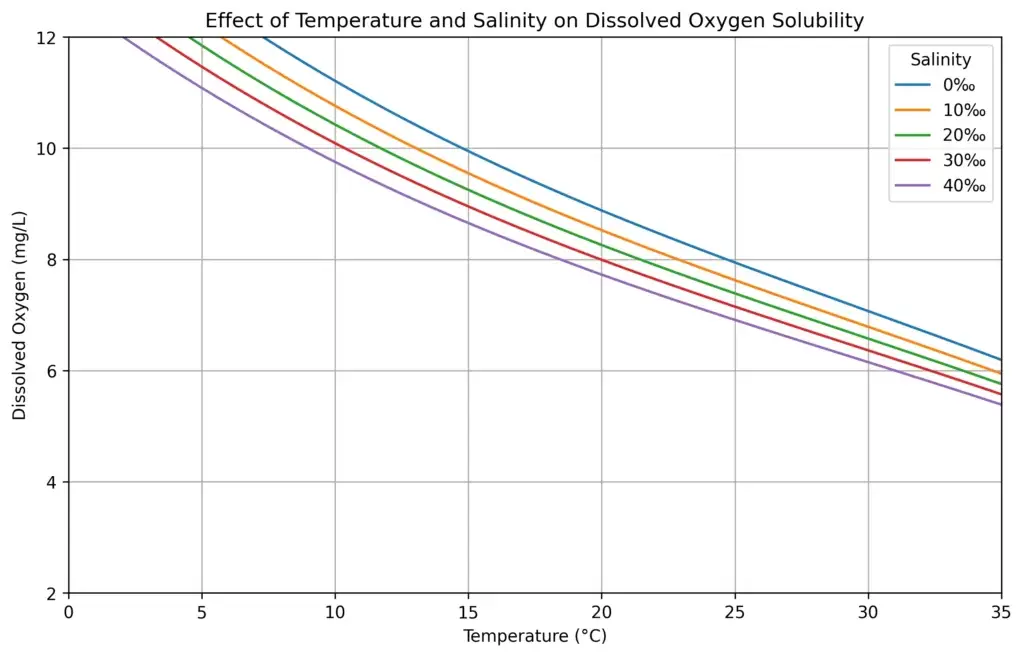
Salinity
Salt in water also reduces the ability of water to retain dissolved oxygen.
As salinity increases, the amount of oxygen that can dissolve in the water gradually decreases. This means marine or brackish systems generally hold slightly less oxygen than freshwater at the same temperature.
| Water Type | Approximate DO Capacity at 20°C |
|---|---|
| Freshwater | ~9.1 mg/L |
| Brackish water | ~8.6 mg/L |
| Seawater | ~8.0 mg/L |
For aquaculture operators working with marine or brackish species, this means aeration and circulation become even more important to maintain safe oxygen levels. The following diagram shows the relationship between salinity and temperature with respect to dissolved oxygen saturation levels.
Atmospheric Pressure
Atmospheric pressure also influences how much oxygen can dissolve in water.
Lower pressure means less oxygen can remain dissolved. This effect is most noticeable at high altitude, but it can also change slightly with weather systems such as low-pressure storms.
In most aquaculture environments the effect is smaller than temperature, but it still contributes to overall oxygen availability
Water Movement and Aeration
Oxygen enters water primarily through exchange with the atmosphere. This process becomes much more efficient when the water surface is disturbed.
Oxygen is introduced into water through:
-
Surface diffusion
-
Mechanical aeration
-
Turbulence and mixing
In natural systems this often occurs when water flows over rocks or cascades in streams.
In managed systems such as ponds, tanks, and raceways, oxygen levels depend largely on mechanical aeration and water circulation.
Balancing Oxygen Replenishment and Oxygen Consumption
Oxygen is added to water through:
- Aerators and diffusers
- Water movement and surface agitation
- Photosynthesis (during daylight hours)
Oxygen is consumed by:
- Fish and aquatic animals
- Microbial activity
- Decomposition of waste
- Uneaten feed
- Biofilters
In heavily stocked systems, oxygen demand can rise very quickly.
The Night-Time Dissolved Oxygen Trap
One of the most common causes of unexpected fish loss occurs overnight.
At night:
- Photosynthesis stops
- Respiration continues
- Oxygen consumption exceeds replenishment
DO levels often reach their lowest point just before dawn — when systems are rarely checked.
This is why systems that appear “fine during the day” can fail overnight.
Aerobic vs Anaerobic Conditions
When dissolved oxygen remains adequate:
- Aerobic microorganisms dominate
- Organic waste is oxidised efficiently
- Water remains stable and low-odour
When DO drops too low:
- Anaerobic organisms take over
- Putrefactive decomposition occurs
- Harmful compounds such as sulphides and amines form
- Water quality degrades rapidly
Maintaining oxygenation protects not only fish, but the entire biological balance of the system.
How Often Should Dissolved Oxygen Be Tested?
Dissolved oxygen levels in ponds, tanks, and natural waterways can change quickly. Temperature shifts, algae activity, aeration performance, and fish respiration can cause oxygen levels to fluctuate significantly during the day.
For most aquaculture and water-quality applications, dissolved oxygen should be checked at least once daily, and more frequently when conditions are unstable.
Many operators measure DO:
- Early in the morning, when oxygen levels are typically at their lowest
- During warm weather, when oxygen demand increases
- After feeding, when fish respiration rises
- Whenever fish behaviour suggests stress
Regular testing allows operators to detect declining oxygen levels before they become dangerous.
In practice, these checks are usually performed using a portable dissolved oxygen meter with a cable probe, which provides stable readings and allows measurements to be taken directly in ponds, tanks, or waterways.
Portable meters allow operators to:
- Quickly check oxygen levels at multiple locations
- Detect developing oxygen problems early
- Confirm that aeration systems are working properly
Because dissolved oxygen responds continuously to factors such as temperature, stocking density, feeding activity, and biological processes, reliable management depends on regular measurement and observing trends over time, not just occasional readings.
How Dissolved Oxygen Is Measured in Practice
In aquaculture and environmental monitoring, dissolved oxygen is typically measured using portable handheld meters with a specialised probe.
These instruments allow operators to check oxygen levels directly in ponds, tanks, or rivers and respond quickly if conditions begin to change.
For occasional testing, a compact dissolved oxygen tester can provide a quick snapshot of oxygen levels.
For fish farms, aquaculture ponds, and regular field measurements, operators typically use a portable dissolved oxygen meter with a cable probe, which provides more stable readings and is easier to use when sampling water.
Portable Dissolved Oxygen Meters from Meascom
Meascom offers portable dissolved oxygen instruments suitable for aquaculture, environmental monitoring and water quality testing.
For occasional testing, the PST-DO30 compact dissolved oxygen tester provides a convenient way to check DO levels.
For aquaculture operators and regular monitoring, the HHT-DO200 portable dissolved oxygen meter provides greater stability and is designed for reliable field use.
View the instruments here:
PST-DO30 Compact Dissolved Oxygen Tester
HHT-DO200 Portable Dissolved Oxygen Meter
Maintaining correct dissolved oxygen levels protects fish health, improves feed efficiency, and prevents costly stock losses.






